Triggered by recently published studies on R&D performance, I have engaged in some interesting discussions over the past few weeks on where to focus productivity improvement efforts. Here in one contiguous article is my take on the key levers for improving productivity in a bioscience R&D organization.
Since the start of 2016, three interesting independent studies have been published on pharma R&D productivity. In January, consultants IDEA Pharma published their sixth annual Productive Innovation Index[1], which (in their words) “ranks biopharmaceutical companies by their ability to successfully bring innovations to market”. Early in February, the consulting firms BCG and KMR published a joint paper in Nature Reviews Drug Discovery entitled: “What drives operational performance in clinical R&D?”[2]. And within a few days of that article, researchers Jack Scannell and Jim Bosley published an excellent academic paper entitled: “When Quality Beats Quantity: Decision Theory, Drug Discovery, and the Reproducibility Crisis”[3], in which they use decision theoretic analysis to show that the dramatic historical decline in pharma R&D productivity was strongly correlated with (in their words), “the progressive exhaustion of predictive models … of clinical utility in man“.
The first study ranks R&D productivity from the standpoint of commercial value added. The second concentrates on operational excellence. While the third examines R&D productivity from the perspective of decision making over the course of an R&D project. All three viewpoints are perfectly valid—what benefits bioscience R&D executives and managers most though is combining them into one complete picture.
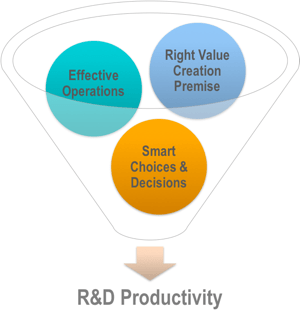
In this article, I describe my personal framework for thinking about R&D productivity improvement, one I have evolved over the years. Not everyone will agree with me, but I hope many of you will find this way of thinking interesting and helpful. I believe there are three major levers for increasing R&D productivity that an organization can pull:
- Right Value Creation Premise.
- Effective Operations.
- Smart Choices and Decisions.
In my experience, optimizing one of these levers alone without consideration of the other two generates at best some short-term benefits, and over the longer-term may cause more harm than good. We’ll start by first clarifying what I mean by “R&D productivity” and then look at each of these levers in turn, before concluding by arguing why they need to be tackled concurrently.
R&D Productivity as Risk-adjusted New Value Creation
At the simplest level, productivity is the difference between outputs and inputs (i.e. “new value created”), sometimes expressed as a financial return on investment ratio. For a single R&D project that is subsequently sold outright, you could say the productivity achieved is the difference between the sale price and the cumulative investment in that project. To increase R&D productivity, you would with this line of thinking, seek to maximize the new value created by every project; measured either on the deal value of the R&D project being partnered out, or the financial net present value of the resulting product sales after launch.

In bioscience R&D, project attrition is often high owing to limited predictability of how the human body responds to new therapeutics and diagnostics. So a more correct way of looking at productivity is to compare the total value created against the total investment made for all projects in your portfolio.
Moreover, high return on R&D investment usually corresponds to high scientific and/or commercial risk (see sidebar: “Four Types of Risk”). Always seeking the highest possible return by taking on the riskiest projects can lead to a disastrous collapse in productivity across the portfolio as all those projects fail.
Your drive to improve productivity must be tempered by the level and kind of risks you are willing to take on. And risk perception varies widely from one organization to another. For instance, although some companies shy away from R&D on Alzheimer’s disease (very low historical success rates, disease etiology not fully understood, expensive long-term clinical trials), others continue to make major investments in this area. Hence R&D productivity improvement must always take place within the constraints of an overall scientific and commercial risk profile acceptable to your top management and owners.
Right Value Creation Premise
When you lead an R&D organization, a very important consideration is having a clear value creation premise[4] i.e. how you set your organization up to create new value. In bioscience R&D, this premise incorporates your views on three fundamental questions:
- Which biological mechanisms, disease areas and technologies will you work with?
- In which parts of the overall R&D chain will you manage projects in? And how will you connect into the other parts of that chain?
- Which aspects of your projects do you (a) conduct mostly in-house, (b) cooperate with strategic external collaborators, or (c) outsource operationally to outside providers?
Answering question 1 involves clarifying the scientific and commercial risk profile that is acceptable to your top management and owners. For instance, in drug development, do you pursue niche indications with high unmet medical needs or mass market “me-better“ products? Or in drug discovery, are you going to gamble on the current immunooncology wave or prospect in the human microbiome?
Whereas questions 2 and 3 relate to how (subject to the constraints of how you answered question 1) you deploy your own resources and capabilities to create new value relative to the capabilities of potential competitors, strategic collaborators and outsourcing providers. For instance in drug discovery, you might in-license small molecule projects with documented but unoptimized lead structures and outsource all your in-vivo pharmacology. Or in drug development you might partner out your projects at clinical proof-of-concept in Phase II and outsource biostatistics while maintaining a strong in-house medical group.
How you answer these questions can have a huge impact on your R&D productivity. But there is no generic best solution—it all depends on your specific circumstances. At any point in time, there is a right value creation premise for you. And the situation is not static. As environmental factors change and you learn more about your own and others’ capabilities, these questions should be re-visited regularly.
Effective Operations
Within the context of your chosen value creation premise, operations is an obvious lever of R&D productivity to tackle. It is the most visible in the short term of the three levers we are discussing here, and also the one that is explicitly budgeted for in the annual financial cycle.
In operations, you manage the classic trinity of cost, speed and quality. Although you should to bear in mind that “quality” has a specific meaning in the bioscience sector, typically relating to GxP standards in Development and experimental reproducibility in Research.
The relative focus on operations by large multinational pharma marketers compared to biopharma SMEs (whether a 5-person research-stage startup or an established 500-person biopharma company) can be somewhat different. A comparatively small percentage improvement in operational performance can, through sheer scale, translate to significant improvement in the financial statements of a large multinational, and often to an even greater corresponding improvement in share price by demonstrating efforts to be efficient with shareholders’ capital. Whereas operational gains for an SME do not have as much impact on enterprise valuation as for example good decisions that shorten time to market or widen the indication label of individual assets even before the products are launched.
An increasingly important factor in operations is outsourcing. You should beware of simply looking for cost savings by working with service providers. Collaborations add collaboration risk (see sidebar: Four Types of Risk") and incur a time and resource overhead (“collaboration tax”) that can reduce speed and create more internal work. As with execution risk, collaboration risk needs to be mitigated as much as possible. Some of the mechanisms for doing so (as described in my book on R&D partnerships) include selecting appropriate collaborators, setting up the collaboration arrangement carefully, and proactively dealing with collaboration issues during the execution.
In a manufacturing organization, productivity and operational efficiency are synonymous. And many of the improvement frameworks used in that domain (e.g. process re-engineering, LEAN workflows, KPIs, strategic sourcing, etc.) have seeped into bioscience R&D improvement initiatives. But these efforts have had varying degrees of success. In manufacturing, the specifications of the products being made are very tightly defined and variability usually reduces value. Whereas in R&D, variability in output compared to the original plan can also have equal or superior value in many cases. Many efforts to optimize bioscience R&D operations do not improve overall productivity and can even harm it, as one drug discovery executive said to me once:
“We have become very fast and efficient at generating placebos."
Hence it is not about the most efficient operations but rather the most effective. Of course you want to conserve resources and save time by doing things right. But you cannot over-emphasize efficiency to the detriment of doing the right things! Which leads us to our final productivity lever to be discussed next.
Smart Choices and Decisions
The R&D journey can be viewed as a series of informal choices and formal decisions that you make for the overall project portfolio and for individual projects. Some of the choices I see many people make are not even conscious but derive from historical precedent and momentum:
I often hear: “We have always done it this way” … to which I often respond: “Haven’t your circumstances changed? Aren’t there any new alternatives available today?"
Your make R&D choices and decisions within the context of your selected value creation premise and subject to the constraints of your overall financial budget. At the portfolio level, you decide which specific projects to work on, and how to balance that portfolio with respect to risk and resource usage. At the individual project level, you choose what to do on every step of the research or development path, including dealing with various unanticipated scientific findings and external environmental changes in real time as they arise. At both levels, you also make choices on how to handle project attrition. In this context, not only is scientific and commercial risk important to address, but also the execution risk (see sidebar: Four Types of Risk) caused by “unknown unknowns”.
The approach taken by large multinational pharma marketers compared to biopharma SMEs can be somewhat different. Consider portfolio choice and attrition management. For a given budget, a big pharma will often manage a large portfolio of high risk high return projects using a “fast and cheap” attrition approach i.e. aiming for each project to reach its failure point as quickly as possible with limited spending, leaving a smaller number of the “fittest” projects to funnel into successive R&D stages.
Whereas an SME usually has access to a much smaller number of scientific assets. Owing to this “scarcity motivation”, it will want to squeeze the maximum value possible out of every asset using its more limited resources. So it might choose to invest in mechanisms of action or molecules that have a wide range of potential applications in multiple disease areas, indications or even diagnostics. This approach maximizes “shots on goal” and increases the chances that something valuable can accrue from the investment. And the path taken for each project might optimize resource utilization by maximizing the information value of each successive experiment.
It’s obvious that optimal choices and decisions derive from good models with high predictive validity. But we’re not always so fortunate as to have the perfect tools available. So it is even more important to have problem-solving and decision-making forums and processes that leverage the most relevant know-how from both your internal organization and existing external collaborators. Furthermore, you need to mitigate execution risk as much as possible. Some of the mechanisms for doing so (as described in my book on R&D partnerships) include selecting appropriate project team members, establishing a strong capacity for creative problem-solving, and putting in place a nimble project management approach, combined with an openness to get help from new unanticipated collaborators as the project progresses.
Concluding Thoughts

These three levers, namely, (1) the right value creation premise, (2) effective operations, and (3) smart choices and decisions, are akin to a trio of musicians. Like the violinist, pianist and cellist in a chamber music trio (or the guitarist, bassist and drummer in a rock band if you’re so inclined), the three combine to produce a complete end result and can stimulate each other to greater heights.
To improve bioscience R&D productivity, you need to address all three levers described in this article. Moreover you need to tackle them concurrently and in an integrated manner as both positive and negative feedback loops exist between them.
For instance, always seeking to conduct the cheapest and quickest possible experimental studies can harm decision-making. Secondly and in contrast, consistently choosing very large and complex “bullet proof“ study designs can dramatically reduce operational performance without necessarily adding much new insight. As a third example, your value creation premise determines what kind of portfolio you choose to have and how you deploy your resources. But insightful information derived from smart experimental choices and learning from your operations can alter your thinking and suggest new and better ways to create value.
References and Notes
-
See announcement on IDEA Pharma’s website: http://www.ideapharma.com/The-Productive-Innovation-Index–2016 ↩
-
Ringel, M., Martin, L., Hawkins, C., Panier, V., Denslow, M., Buck, L. and Schulze, U. 2016. “What drives operational performance in clinical R&D?” Nature Reviews Drug Discovery published online February 5, 2016. http://www.nature.com/nrd/journal/vaop/ncurrent/full/nrd.2016.2.html ↩
-
Scannell, J.W. and Bosley, J. 2016. “When Quality Beats Quantity: Decision Theory, Drug Discovery, and the Reproducibility Crisis” PLOS ONE published online February 10, 2016 http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0147215 ↩
-
I used to call this “R&D Strategy”, but the “strategy” word is so over-used these days that now I prefer a phrase which is more specific. ↩


